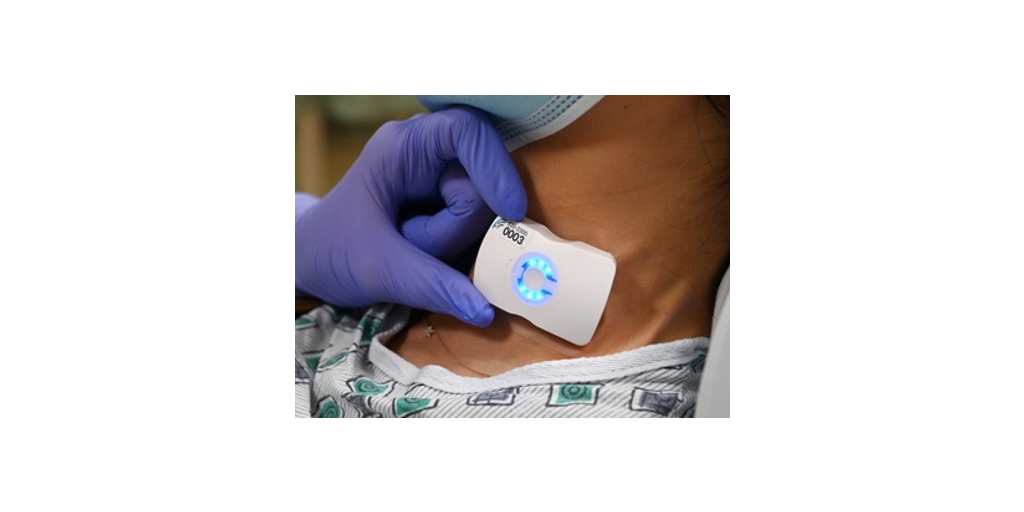
Contract recognizes FloPatch’s unique potential to improve fluid management decisions and patient outcomes in sepsis care
LOS ANGELES–(BUSINESS WIRE)–#flopatch–Flosonics Medical announced that its FloPatch wearable Doppler ultrasound device has received an Innovative Technology contract from Vizient®, the nation’s largest provider-driven healthcare performance improvement company. The contract was awarded following review by hospital experts serving on one of Vizient’s client-led councils, who identified FloPatch’s unique potential to improve fluid management decisions and patient outcomes in sepsis care.
Innovative Technology contracts are recommended after review and interaction with products submitted through Vizient’s Innovative Technology Program. Vizient’s client-led councils identify technologies that have the potential to enhance clinical care, patient safety, healthcare worker safety, or improve the business operations of healthcare organizations. If a product is determined to be innovative, Vizient may award a contract outside of the competitive bid cycle.
“FloPatch was designed to give frontline clinicians the hemodynamic data they need to make better fluid decisions, faster, and without additional specialized training or equipment,” said Joe Eibl, CEO and co-founder of Flosonics Medical. “This Vizient Innovative Technology contract validates what hospitals have experienced firsthand: that bringing objective, real-time fluid responsiveness data to the bedside can transform sepsis care delivery. Through this contract, we’re excited to bring enhanced access of this technology to patients who need it.”
Addressing a Critical Gap in Sepsis Care
FloPatch is the first FDA-approved, wireless, wearable Doppler ultrasound device designed to assess blood flow. These assessments are used to guide personalized intravenous (IV) fluid management for patients with sepsis and other critical conditions. Research shows that nearly one-third of patients with septic shock do not respond to additional IV fluids¹, yet clinicians have historically lacked efficient, scalable tools to assess fluid responsiveness at the bedside.
The clinical evidence is compelling: dynamic fluid assessments have been shown to reduce the need for mechanical ventilation and renal replacement therapy²³, decrease ICU length of stay³, and are associated with lower mortality⁴. FloPatch addresses this gap by enabling frontline clinicians to quickly understand a patient’s fluid responsiveness in minutes, providing objective, real-time hemodynamic data that helps guide when more fluid is needed and when it is not.
Scalable Across Health Systems
Unlike traditional hemodynamic assessment approaches that require additional specialized training, dedicated equipment, or expert interpretation, FloPatch is designed to be used by any clinician at the bedside. This makes it uniquely scalable across emergency departments and intensive care units, enabling health systems to standardize and elevate sepsis care delivery across their entire organization while supporting compliance with sepsis care protocols, including CMS SEP-1 requirements.
“After a review of the FloPatch Wearable Doppler Ultrasound, Vizient’s client-led council agreed this technology offers a unique benefit over other products available in the market today and recommended it for an Innovative Technology contract. We are pleased to award this new contract to Flosonics,” said Kelly Flaharty, associate vice president, contract operations for Vizient.
Vizient represents a diverse client base that includes academic medical centers, pediatric facilities, community hospitals, integrated health delivery networks, and non-acute healthcare providers, with a contract portfolio representing more than $156 billion in annual purchasing volume. Through its Innovative Technology Program, Vizient works with client-led councils and task forces to evaluate products for their potential to bring meaningful innovation to healthcare delivery.
About Flosonics Medical
Flosonics Medical is a medical device company engaged in the research and development of innovative ultrasound technology. Founded in 2015, the company’s mission is to improve patient care and the practice of medicine through technology-enabled solutions and groundbreaking clinical research. For more information, visit www.flosonicsmedical.com.
| References |
| ¹ Hernández G, Ospina-Tascón GA, Damiani LP, et al; ANDROMEDA-SHOCK Investigators. Effect of a resuscitation strategy targeting peripheral perfusion status vs serum lactate levels on 28-day mortality among patients with septic shock: the ANDROMEDA-SHOCK randomized clinical trial. JAMA. 2019;321(7):654–664. |
| ² Douglas IS, Alapat PM, Corl KA, et al. Fluid Response Evaluation in Sepsis Hypotension and Shock: A Randomized Clinical Trial. Chest. 2020;158(4):1431–1445. |
| ³ Latham HE, Bengtson CD, Satterwhite L, et al. Stroke volume–guided resuscitation in severe sepsis and septic shock improves outcomes. J Crit Care. 2017;42:42–46. |
| ⁴ Dubin A, Loudet C, Kanoore Edul VS, et al. Characteristics of resuscitation, and association between use of dynamic tests of fluid responsiveness and outcomes in septic patients: results of a multicenter prospective cohort study in Argentina. Ann Intensive Care. 2020;10(1):40. |
Contacts
Media Contact:
Jamie Gray, Flosonics Medical, [email protected]